AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Sulfate ion bonding9/20/2023  
It was a bit of work, but we have the best structure here. We add our 2 minus right there.Īnd that is the Lewis structure for SO4 2. 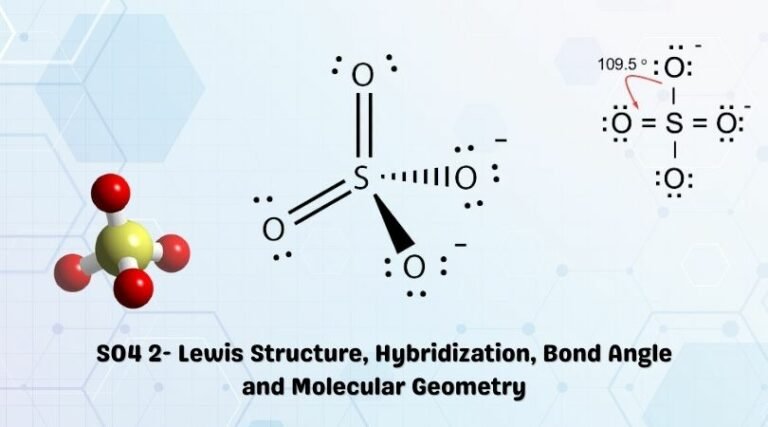
We need to put brackets around it to show that it's an ion and the charge of the ion. Since it's an ion, there's one last thing we need to do. Since more of the formal charges are zero, this is a better structure for SO4 2. But if you look, you have a negative 1 and a negative 1 here. So at this point, we see that we have mostly zeroes. Finally, looking at the blue Oxygens, we have 6 minus 6 nonbonding, and then 2 bonding divided by 2. If we look at the green Oxygens, you can see that we have 6 minus 4 of the nonbonding, and then 4 bonding we divide by 2. So the formal charge on the Sulfur is zero. So for Sulfur, 6 minus zero, there are no nonbonding and now we have 2, 4, 6, 8, 10, 12 total bonding electrons. Let's see how that changes the formal charges. So I've moved electrons from the outside of these two green Oxygens into the middle to form double bonds. Let's try that and then recalculate our formal charges. If I do that twice, if I move these two into the middle to form double bonds, and get rid of them, I think that'll get rid of the positive 2 charge. When I see this +2 charge here on the Sulfur, I know that I can move electrons from the outer valence electrons of the Oxygen into the middle. So let's see if we might be able to do another Lewis structure that has more zeroes for the formal charges. That does make sense, but with formal charges, we want them to be as close to zero as possible for the atoms. If we add up all the formal charges, the -1, -1, -1, -1 and +2, we do get a total charge of negative 2. Six minus 6 minus 2 gives us a minus 1 formal charge for each Oxygen. And all these Oxygens are the same, so we only need to do one. Up here, nonbonding, we have 6 and then bonding, we have 2. For the Oxygen, it's also in group 6 or 16, so it has 6 valence electrons. Six minus zero minus 4 gives us a +2 formal charge for the Sulfur. And the bonding electrons, 2, 4, 6, 8 we've used 8 of those, and we'll divide that by 2. 
Up here, all of the electrons, all of them are involved in bonds, so that's going to be zero. So to calculate the formal charge on the Sulfur: we see that Sulfur, on the periodic table, group 16 or 6, has 6 valence electrons. So we really do need to check our formal charges. That means it can hold more than 8 valence electrons. Sulfur is in the third period of the periodic table. Looking at the structure here, we see that each of the Oxygens has 8 valence electrons 2, 4, 6, 8 as does the Sulfur here, 2, 4, 6, 8. Let's go around the outer atoms and make sure they have octets. Next, we'll draw bonds between the Sulfur and the Oxygens, so there we have four bonds and we've used eight valence electrons. We'll put the Sulfur in the center, and then the four Oxygens will go on the outside. That gives us a total of 32 valence electrons. On the periodic table: Sulfur, 6 valence electrons Oxygen also has 6, we have 4 Oxygens, multiply by 4 and these 2 valence electrons up here, we need to add those, as well. Let's do the SO4 2- Lewis structure, for the sulfate ion. Thus the representation sulfate ion in the form of S=O is better. The single bonded oxygen atom remains have same formal charge i.e -1in both representation. The formal charge for the double bonded oxygen atom increases from -1(represented with four single bond) to to 0 in the representation with two double bonds (represented with two double bonds). For sulphur, the formal charge in the single bond representation is +2 while in the double bond it decreases to zero. The representation sulfate ion in the form of S=O is better because it minimizes the formal charge. The representation of sulfate ion with S-O bond The representation of sulfate ion with two S=O bondįormal charge of O (single bond) = 6-(6 +½ (2)) = -1įormal charge of O (double bond) = 6-(4 +½ (4)) = 0 (a) Here we have to describe which representation is better from the standpoint of formal charges.ġst we will draw the two ways of representation of sulfate ion, then we will calculate their formal charge. The sulfate ion can be represented with four S-O bonds or with two S-O and two S=O bonds. There are no lone pairs of electrons on the S atom for SO4 2- (Sulfate ion). 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
